Long COVID refers to a range of persistent symptoms that can linger for weeks, months or years after...
Read More
Dr. Gregory Breen, MD is a board-certified physician in pulmonary, critical care and sleep medicine. He has played an instrumental role in Inspira's administration of outpatient monoclonal antibody therapies for qualifying patients with COVID-19.
What is antibody therapy?
The concept of antibody therapy, or what's called "passive immunity," has been around for over a hundred years. Similar types of treatment have been used for measles, rabies, tetanus, hepatitis and influenza. In fact, even the Spanish flu, the great pandemic prior to this one, antibody therapy was used in that situation as well. So this is a longstanding therapy that has been promising in several disease processes.
How does monoclonal antibody therapy differ?
The difference with monoclonal therapy is that we are isolating a particular antibody that is most effective against this disease. And that is a unique concept. Not only are we isolating it, but it's being prepared in a laboratory—it is a pure form of the treatment. This avoids all of the pitfalls of transfusing plasma or blood from a convalesced patient. So, the monoclonal antibodies are a single, meaning "mono," clone, or line of antibodies—a single type of antibody— that is then targeted against a part of the virus. Typically, the spike protein is one of the main components of the virus.
Talk about the efficacy of monoclonal antibody treatments for patients with COVID-19.
The treatment is extremely effective. And in fact, phase three studies have shown that the treatment can decrease your chances of being hospitalized or dying from COVID by about 70%. So you're sicker for a shorter period of time and you have a much less chance of being hospitalized or dying from the disease if you receive this treatment.
How is this type of treatment administered?
The key is to get the monoclonal infusion before you become very symptomatic and before you require hospitalization. The infusion takes about an hour and then we observe for an hour, but it's a very, very relaxed atmosphere where you can get some rest, which you typically need when you have COVID. And thankfully, because of our collaboration between our primary care, urgent care and emergency room physicians, and the great support from the in Inspira administration and nursing, we are able to do the transfusions on a daily basis, Monday through Friday, at our medical centers in Mullica Hill and Vineland—and we've actually treated over 200 patients with this therapy successfully.
I was just diagnosed with COVID-19. Amy I eligible for this?
These treatments have been approved under an emergency use authorization from the government. There are strict criteria that have to be followed before we can administer them to a patient-—and they fall into three categories.
What I can I expect after receiving the treatment?
Side effects have been extremely rare. You can have nausea, flushing, a little bit of a fever, potentially, during the infusion. You may even have an allergic reaction to the infusion, but again, those have been exceedingly rare and allergic reactions in our 200+ patients treated here at Inspira, we have not seen one of those. This is not necessarily a symptom based treatment, meaning, you may not feel better the next day after the infusion. The idea here is that, in the first week of COVID you are suffering from a lot of flu like symptoms, generally fevers, nausea, muscle aches and fatigue. In the second week of COVID, the life-threatening manifestations can occur. That can lead to hospitalization and death. The monoclonal therapy is meant to prevent that second week complications, prevent you from being hospitalized and prevent you from dying from the disease. So even if you don't feel a difference with the treatment, it is helping you by getting rid of the virus quicker from your system, preventing it from binding to yourself, allowing your body to eliminate it and thus prevent the complications by about 70%.
Any thoughts as we look to the future of this pandemic?
As we approach this next phase of the pandemic, we are still seeing surges in cases and they are primarily being driven by some of the mutant strains that we've seen. And so it's so important, even as we look towards the future in a hopeful way, that we maintain our vigilance and that we continue to follow the guidelines from the government and our health care professionals, including wearing masks, keeping social distancing and washing hands. What I can tell you as a practicing physician and as someone who has really kept up with this throughout the past year, I've just been impressed by the miracle of the vaccines. There were no steps skipped. They were done with rigorous scientific study and with the help of governments and industry collaborating together in a way that we've never seen before. So to me, these are a miracle treatment, a lifesaving therapy that I really encourage all of my patients to consider. That being said, we are all individual human beings and we must make the best decisions for ourselves and our families. And I encourage patients to discuss this issue with their physicians at every opportunity. Physicians will have a little bit more of an understanding of some of the science and be able to reassure patients that the science is sound and that there were no steps skipped in the development of the vaccines.

Long COVID refers to a range of persistent symptoms that can linger for weeks, months or years after...
Read More
A Very COVID Christmas: How to Celebrate Safely
Read More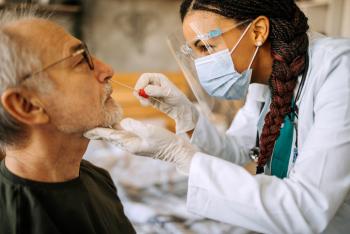
After more than two years of masks, social distancing and travel restrictions, this winter is likely...
Read More
The material set forth in this site in no way seeks to diagnose or treat illness or to serve as a substitute for professional medical care. Please speak with your health care provider if you have a health concern or if you are considering adopting any exercise program or dietary guidelines. For permission to reprint any portion of this website or to be removed from a notification list, please contact us at (856) 537-6772